What Are Renú® Voice And Renú® Gel?
Renú® Voice and Renú® Gel are both injectable implants. Renú® Voice contains microspheres of calcium hydroxylapatite (CaHA), a biocompatible material that is suspended in a hydrogel carrier. Renú® Voice and Renú® Gel are both designed to replace volume lost in the tissue of vocal folds. These injections assist in bringing the folds closer together to aid in phonation and the restoration of the voice for short-term (Renú® Gel), and long-term (Renú® Voice) non-permanent restoration and augmentation.
What Will Be Experienced During And After A Renú® Voice And/Or Renú® Gel Procedure?
In most cases, the Renú® Voice and/or Renú® Gel procedure will be performed in a physician’s office or treatment clinic. Patients will be made comfortable with the use of either a topical or local anesthetic for the treatment. This process takes about 15 – 20 minutes. During which time patients may be asked by their doctor to make sounds to assess the improvement of their vocal quality.
For Renú® injection procedures, there is no traditional recovery period. Your physician may ask you to refrain from talking post-procedure and suggest voice rest for a few days. This will vary based on patient’s condition and treatment needs. Few side effects have been reported and those that are reported tend to center on side effects associated with injected implants. Tenderness and mild swelling at the injection site are commonly reported.
Is Surgery A Better Option?
Only a patient’s physician should make the evaluation and recommendation for surgery. Injection therapy using Renú® Voice and Renú® Gel can be completed as an in-office or surgical procedure where local anesthesia is not required, but may be utilized at the injection site. Delivery of treatment can be completed percutaneously or transorally. If patient’s physician determines that surgery is required, it does demand that patients go to an operating room and receive general anesthesia with a recovery period.
What Are The Next Steps?
Schedule time to talk to a physician about Renú® Voice and/or Renú® Gel to see if vocal fold injections are the right option.
Where Can Renú® Voice and Renú® Gel Be Purchased?
Contact Cytophil, Inc. by email: orders@cytophil.com or calling toll free: +1 855-433-2765
Please include a contact name, email address, and phone number with your inquiry.
Does Renú® Voice and Renú® Gel Harden After Injection?
The short answer is no. Since osteoblasts and osteoclasts do not exist above the periosteum, there are no catalytic cells to stimulate hardness.
Should The Patient Expect Any Side Effects?
Few side effects have been reported and those that are reported tend to center on side effects associated with injected implants. Tenderness and mild swelling at the injection site are commonly reported.
How Long Will Renú® Voice And Renú® Gel Last In Patients?
Renú® Voice is comprised of two main components, each dissipating over different periods of time. The quickest to be absorbed is the carrier gel which takes a few weeks. Next the CaHA particles are broken down gradually into calcium and phosphate ions metabolized by the body’s natural process over a few months. Thus, Renú® Voice should provide patients long-lasting results for non-permanent restoration and augmentation of vocal folds. Typical Renú® Voice vocal fold implant can be expected to last from 9 to 18 months.
Renú® Gel is a hydrogel, that is quick to be absorbed by the body, which takes a few weeks. Thus, Renú® Gel should provide patients shorter-term results in comparison to Renú® Voice, for non-permanent restoration and augmentation of vocal folds. Typical Renú® Gel vocal fold implant can be expected to last from 10 to 14 weeks.
Does Renú® Voice And Renú® Gel Require Special Storage?
No. Renú® Voice and Renú® Gel may be stored at ambient room temperature between 59°F and 90°F (15°C and 32°C). Renú® Voice and Renú® Gel may be stored if packaging is intact and undamaged for up to two years.
REMINDER: Renú® Voice and Renú® Gel are manufactured as a single-use product and once opened, should not be stored or re-used.
How Is Renú® Voice And Renú® Gel Packaged?
Renú® Voice and Renú® Gel come in a sterile foil pouch containing a single-use, pre-filled syringe inside a protective outer box. Renú® Voice and Renú® Gel syringe have a packaged volume of 1.5cc. Upon opening the foil pouch it is common to find water droplets that are consistent with the normal sterilization process. Renú® Voice and Renú® Voice are manufactured as a single-use product and any post-procedure product remaining in the syringe should not be stored or re-sterilized for future use.
How Does A Patient Know If They Need To Consider Treatment For Their Voice?
Voice issues sometimes occur but if a patient is experiencing symptoms that persist or even seem to be getting worse, treatment may be required.
- A weak or airy quality to voice
- Difficulty or inability to increase voice volume
- Difficulties in breathing and/or swallowing
- Difficulties in swallowing resulting in choking
- Persistent feeling of something in throat
Symptoms like the ones described above could be an indication of other health concerns including the possibility of throat cancer. Because these issues may have serious health implications, patients should see your physician for evaluation.
What Physician Should A Patient See For Persistent Issues With Their Voice?
An Ear, Nose and Throat (ENT) physician, also know as otolaryngologist, will conduct an exam to evaluate whether the patient’s voice issues are of a serious nature and need medical treatment. This process will include exploring the patient’s medical history and performing a physical exam of their vocal folds. Vocal fold examination can be as simple as looking into the patient’s throat with a small mirror and may also include looking closer with a camera tube that is passed through the nose known as an endoscope. If an endoscope is used, the throat is numbed to provide patient comfort during the vocal exercises so the physician can see the patient’s vocal folds in motion.
If Treatment Is Needed What Choices Does The Patient Have?
If the patient’s medical exam resulted in insufficient vocal fold performance, then their physician may consider one or more treatment choices.
- Seeing a speech-language pathologist to assist in better voice control
- Surgery may be indicated to move the vocal folds closer together
- Injection therapy using Renú® Voice and/or Renú® Gel to bring the vocal folds closer together for non-permanent restoration and augmentation.
- Treatment can be completed as an in-office or surgical procedure where local anesthesia is not required but may be utilized at the injection site.
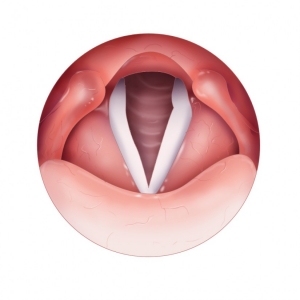
Vocal Fold Inspiration
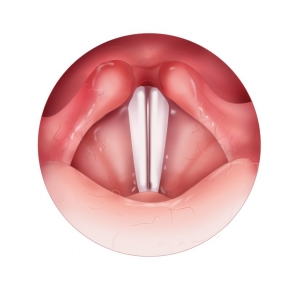
Vocal Fold Phonation
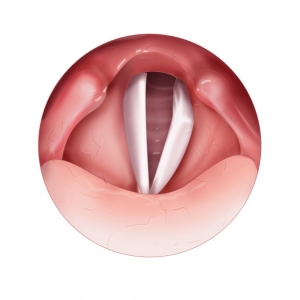
Attempted Phonation
with Compromised Vocal Fold
Rosen, C.A., J. Gartner- Schmidt,(et al.), “Vocal fold augmentation with calcium hydroxylapatite (CaHA). Otolaryngology Head and Neck Surgery (2007) 136, 198-204
Rosen, C.A., J. Gartner- Schmidt,(et al.), “Vocal fold augmentation with calcium hydroxylapatite (CaHA): Twelve month report. Presented on May 19, 2006 at the Annual Meeting of the American Bronco-Esophageal Association, Chicago, IL.
Rosen, Clark A. (et al.), “Vocal Fold Augmentation with Injectable Calcium Hydroxylapatite: Short-Term Results.” Journal of Voice, September 2004, Vol. 18, No. 3. p. 387-391.
Belafsky, Peter C. (et al.), “Vocal Fold Augmentation with Calcium Hydroxyapatite.” Otolaryngology – Head and Neck Surgery, October 2004, Vol. 131, No. 3, p. 351-354.
Chhetri, Dinesh K. (et al.), “Injection Laryngoplasty with Calcium Hydroxylapatite Gel Implant in an In-Vivo Canine Model.” Annals of Otology, Rhinology & Laryngology, April 2004, Vol. 113, No. 4, p. 259-264.
Hughes, Richard G. M., “Vocal Cord Medialization by Transcutaneous Injection of Calcium Hydroxylapatite.” Journal of Voice, December 2005, Vol. 19, No. 4, p. 674-678.
Kwon, T. K. (et al.), “Preliminary Results of a New Temporary Vocal Fold Injection Material.” Journal of Voice, December 2005, Vol. 19, No. 4, p. 668-673.
Note: These studies were done with Prolaryn™ Plus or Prolaryn Gel (Formerly Radiesse® Voice or Radiesse gel, Merz) but are relevant since Renu® voice and Renu® gel are substantially equivalent products and contain the same components in the equivalent formulations. Radiesse® and Prolaryn™ are registered trademarks or trademarks of Merz North America.